The K factor is an estimate of the dissemination of a particular infection. In day to day use, it gives an idea of how closely a disease clusters vs. disperses throughout the population. This could be a result of how easily it infects people or how much an infectious agent, like a virus, is scattered from an infected person to a second person, then beyond. The lower the K factor, the more a disease tends to cluster in small groups of people rather than disperse throughout the population. This means that it may infect a close group of people, but does not have secondary or tertiary spread.
Most coronavirus infections are thought to have low K factors, with estimates for SARS being 0.16 and MERS being about 0.25. This is obviously important because SARS and MERS tended to have minimal dispersion beyond the initially infected individuals, being contained within tight population clusters, and as a result died out. In contrast, influenza has a relatively high K value of 1 or greater, suggesting that it distributes widely through the population and does not cluster.
It is therefore important to identify the K factor for COVID – 19. Will it be limited, like other coronaviruses, or have broader spread? And if it has a small K factor, how could it have caused such an extensive “first wave?”

An excellent review in Science Magazine recently addresses this issue. The article noted that the R value (subject of my blog post last night) is lower than initially thought for COVID. But the K factor may actually be more important. In a March preprint, Adam Kucharski, Associate Professor, London School of Hygiene & Tropical Medicine, estimated the K factor for COVID is only 0.1. “Probably about 10% of cases lead to 80% of the spread,” Kucharski says. This means that the infection is not easy to catch and it does not spread beyond the first group of people infected. It eventually dies out on its own.
Supporting this concept is the “super-spreader” phenomenon observed with COVID-19. Superspreading happens when one event and potentially a single person infects many others. The most well studied of these events involved a choir practice in Mt. Vernon, Virginia. A single infected person infected 53 choir members, three were hospitalized, and two died. Many other events have been identified. A database by Gwenan Knight and colleagues at the London School of Hygiene & Tropical Medicine (LSHTM) lists an outbreak in a dormitory for migrant workers in Singapore linked to almost 800 cases; 80 infections tied to live music venues in Osaka, Japan; and a cluster of 65 cases resulting from Zumba classes in South Korea. Clusters have also occurred aboard ships and at nursing homes, meatpacking plants, ski resorts, churches, restaurants, hospitals, and prisons.
If this is true, and this disease spreads only tightly in clusters, how did the “first wave” of the coronavirus infection occur?
There are several common factors involved with these events. First, they usually happened indoors. Researchers in China studying the spread of the coronavirus identified 318 clusters of three or more cases between 4 January and 11 February, only one of which originated outdoors. A study in Japan found that the risk of infection indoors is almost 19 times higher than outdoors. Some situations may be particularly risky. Meatpacking plants are likely vulnerable because many people work closely together in spaces where low temperatures help the virus survive according to the Science review.

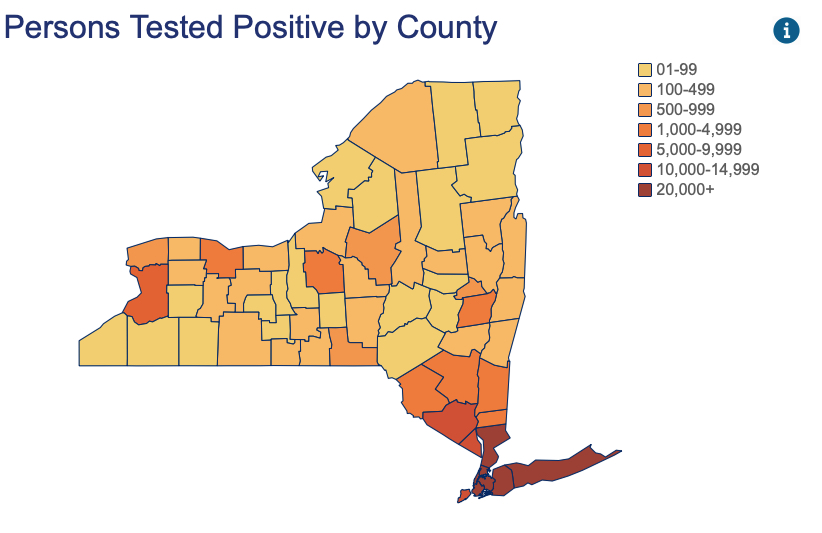
This could be explained if the first “COVID-19 wave” was not so much a wave as a number of very large clusters. There were massive clusters in places like New York City where there is a very high population density with many opportunities for super spreading. Interactions with many infected people on subways, offices and housing units and social establishments (bars!) could all be characterized this way. They also had many nursing homes with multiple highly susceptible individuals who were inadvertently seeded by placement of infected patients.

On the other hand, this could also explain why places like Florida have had limited cluster outbreaks other than in nursing homes. It could be because people spend more time outside, the outside temperatures are much warmer, and UV light is abundant.
Given a low K value for COVID, what does the clustering of this disease mean moving forward? The first concept is that new infections will probably only occur in clusters. These clusters would be similar to the reports of prior clusters, which almost entirely occurred indoors. In addition, they tend to involve lower temperatures and damp areas, which would be more optimal for viral transmission. They also would require participation of susceptible individuals who would be more easily infected.
If future outbreaks would likely be in clusters, this means they could be limited if certain protections were put in place. These would include making sure that particular care is taken to separate individuals indoors. There has been no large outbreak facilitated out-of-doors, and that would be less likely to yield large clusters. Outdoor gatherings with adequate respiratory spacing should be particularly encouraged. In contrast, in situations where cluster opportunities are abundant, such as nursing homes and meatpacking plants, precautions are necessary to shield residents and make sure that they remain safe.
If clustering makes a second wave across the nation much less likely, it does suggest that some clusters will occur. These may be more frequent in the winter when people are again indoors and temperatures are lower. Patients in nursing homes and other cluster enhancing environments need to be protected. But knowing about clustering and the important role it plays in spreading infection suggests that if cluster conditions could be avoided, we will not see large outbreaks of infection impacting the population.
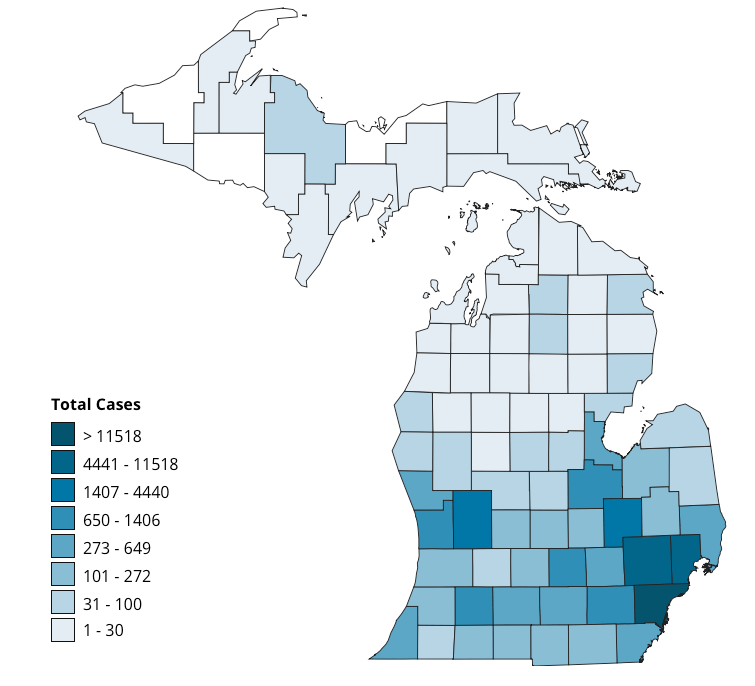
The future should look more like Florida than NYC, and more like Denver than Detroit. Importantly, it should be manageable without shutting everything down again.