
The Moderna SARS-CoV-2 vaccine (identified as mRNA-1273) has received much notoriety as it was one of the first to begin testing. In addition, there was a fair amount of criticism when several of the company’s senior corporate officers and investors sold stock in the company soon after a rise in the stock’s value due to a press release suggesting success of their phase one trial (but offering no data).
Today, we finally saw the data from the first clinical trial as it was published in The New England Journal of Medicine. This vaccine works by using RNA that codes for the viral spike protein. The RNA is injected and taken into human recipient cells that then make the protein. The individual’s immune system then makes a response to this protein. Forty-five individuals were immunized in three dose groups (25, 100 and 250 ug of RNA coding for the spike protein of the virus). Each individual was vaccinated twice, with the second shot “boosting” the immune response.

Two things were evaluated in the Phase I trial. The recipients were evaluated for adverse events from the vaccine and immune responses to the viral protein and virus. Overall, the results are encouraging but preliminary and there is no data that unequivocally suggests this vaccine will ultimately be successful. The publication’s conclusion was that development of the vaccine should continue because it “induced anti–SARS-CoV-2 immune responses in all participants, and no trial-limiting safety concerns were identified.” Despite this optimism, there are some caveats.
The immune responses to the SARS-CoV-2 virus protein were significant and included high titers of antibodies to the virus that were able to inhibit viral growth in tissue culture cells. These so-called “neutralizing antibodies” are thought to be important in the anti-viral response. There were also T cell responses that were indicative of immunity that could destroy virally infected cells. The immune responses elicited by the vaccine were impressive and, in many cases, greater were greater than those induced in individuals who had natural infection.
While there was no serious adverse event that stopped the trial, such as the death or disability of a participant, there were a number of severe adverse events, especially in the highest dose (250ug) group after the second immunization. Systemic adverse events (fevers, aches, muscle pains) occurred in 7 of 13 participants (54%) in the 25-μg group, all 15 in the 100-μg group, and all 14 in the 250-μg group, with 3 of those participants (21%) reporting one or more severe events.
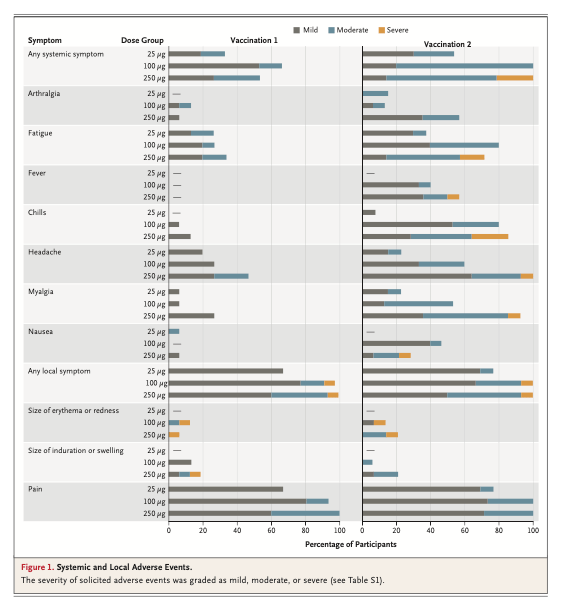
Local adverse events, when present, were mostly mild or moderate, with pain at the injection site being most common. Looking at both the first and second vaccinations, adverse events that occurred in more than half the participants included fatigue, chills, headache, muscle pain, and pain at the injection site. The adverse events were significant enough that the highest, 250 ug dose will be dropped. Fortunately, the immune response to the 100 ug dose appeared adequate.
For an initial clinical trial, the data are very encouraging. Importantly, the immune responses demonstrated did not appear to be able to “enhance infection”; in other words make COVID-19 worse. There will be other difficulties with this vaccine, such as storage and distribution problems, given it is RNA. If the vaccine does work, however, those will be minor issues.
I found 2 reasons to be skeptical of this vaccine news:
1) Adverse reactions were experienced in the trial group (of healthy 18-55 year olds), such as 103 degree fevers, etc. How can this be given to millions of elderly or those with pre-existing conditions, without doing a lot of harm? Typically vaccines given to millions don’t show adverse reactions such a significant % of patients. No data for the most vulnerable population (>55yo).
2) Antibodies were only produced after the 2nd shot was given, and faded after a month (neutralizing antibodies declined from day 43-57). Poor T-cell responses, esp CD8. So it’s unclear if this offers any lasting immunity (or if vaccine/booster will have to be given repeatedly every couple of months into perpetuity?)
Who knows, perhaps the FDA will just jam it through and unleash it onto hundreds of millions of people, but based on this data, I’m not sure I would be comfortable taking it, let alone recommending it to my parents or any elderly folks!!
LikeLiked by 1 person
I agree given the current results. There is always a trade off between reactogenicity (side effects) and protection. I actually think it may be less reactive in the elderly, but also even less likely to be effective. Thanks for the comment!
LikeLiked by 1 person