Two companies are beginning large scale clinical trials of their COVID-19 vaccine candidates. Moderna has begun their large Phase III trial using their mRNA vaccine. It is focused on two doses (shots) of 100 micrograms each. This was the middle dose from the first clinical trial, which produced stronger immunity then the lowest dose. The side effects were less than the unacceptable illness and fevers seen with the highest dose, 250 milligram per shot.
Pfizer and BioNTech are also starting a large phase III study of their mRNA vaccine candidate for Covid-19. Of interest, it is not the one that they recently reported in their original clinical trial. That trial used a vaccine candidate called BNT162b1 that produced only a portion of the spike protein, which SARS-CoV-2 uses to enter cells. The new vaccine called BNT162b2 produces an “optimized” version of the whole spike protein, which Pfizer said in a statement “should lead to more consistent immunity across diverse populations and in older adults.” I have no idea why they might think this, and there is no published data explaining why they changed vaccines after their first trial.
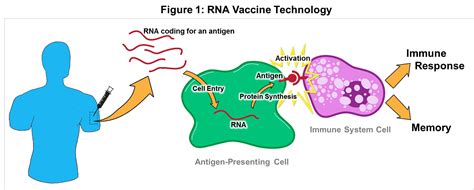
There are many similarities between these two trials. Both are large trials of approximately 30,000 subjects designed to determine if the vaccines can reduce the rate of infection with COVID-19 by 50%, the threshold that the FDA commissioner has determined would be acceptable efficacy. Both vaccines are spike protein (S), mRNA based, meaning the RNA coding for the coronavirus S protein is injected into the individual whose own cells then make the protein. This is a totally new approach to vaccines that has never been shown to be successful in humans.
If anyone is interested in volunteering, NIH has put out an announcement of how to volunteer, and the website is active now. Many sites around the country will participate, but with different vaccines. For those local to Ann Arbor, UM may be testing the Astra Zeneca/Oxford vaccine.
Also, a totally different issue related to COVID-19 vaccines arose today. Over the past 30+ years the Advisory Committee on Immunization Practices (ACIP), has been the primary panel making recommendations on vaccination policy to the Centers for Disease Control and Prevention. They make important decisions such as the dose and timing of all childhood vaccines and also select influenza vaccines for each winter.
Last Friday, however, NIH and the National Academy of Medicine named an expert panel to determine who should be vaccinated first for COVID-19. This should be particularly at-risk individuals, such as the elderly, although as of yet these populations have not been included in the vaccine trials.
In addition to these two committees there is Operation “Warp Speed,” the government’s vaccine production committee. It also claims responsibility for decisions on the distribution of COVID-19 vaccines. Therefore, there are now three committees responsible for this one task.

