It is next June. Several of the COVID-19 vaccines in development have been shown to be effective. Tens of millions of doses of these vaccines have been produced, much of it made even before the efficacy studies were finished to save time. These vaccines are now ready to be distributed to at-risk individuals throughout the country. However, most Americans can’t get the vaccine. Why has this happened?
The limiting factor for getting vaccines to citizens across the country will likely not come from the vaccines themselves but through problems in their storage and distribution. Many of these vaccines are new technologies that require unique storage requirements. In addition, the storage necessities will make it very difficult to distribute these vaccines through sites that normally provide them to people, such pharmacies.
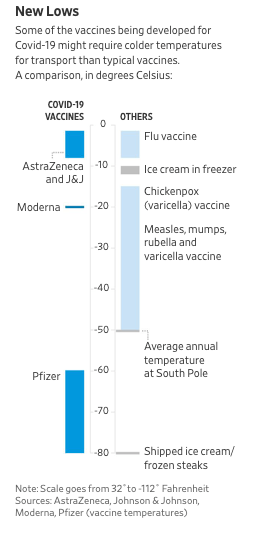
Two of the Covid-19 vaccines better farthest along in clinical development, from Pfizer Inc. and their partner BioNTech SE and from Moderna, rely on a genetic material called mRNA. RNA is inherently unstable and requires the vaccines be stored at very low temperatures. In current studies, these vaccines have been stored at ultracold temperatures, around minus 70 or minus 80 degrees Celsius. This is really cold and freezers that can achieve these temperatures are not readily available in most doctors’ offices or your local CVS/Walgreens.
These temperatures also require special containers and a source of extreme cold (usually dry ice) for shipping. Even short disruptions that allow the temperature to rise for a few hours renders the vaccines useless. The ability to ship and store vaccines at these low temperatures is called the “cold chain.”
While the ability to distribute these vaccines in an appropriate “cold chain” does not currently exist at the scale needed, several companies are working on improving their distribution systems. McKesson reports they will be able to ship Covid-19 vaccines requiring refrigeration to minus 20 Celsius. They are also working with Cryoport, which provides temperature-controlled shipments for livestock-animal vaccines at minus 196 degrees.
United Parcel Service is constructing freezer farms filled with mobile freezer units in Louisville, Ky., and the Netherlands to serve as storage facilities during distribution. Despite this, the distribution chains necessary for these vaccines are still not likely to be adequate for the number of doses necessary.
A final problem is the number of needles needed to inject these vaccines. It is estimated the number of needles available in the supply chain may not be adequate and will cause shortages throughout the health care system.
Therefore, making the approved vaccine for COVID-19 may be the easiest part of the process of immunizing the population!
Meds in except Mycelex
Blood still going in slowly- so far so good
Will take M after am less tied down
Used urinal at bedside☑️300
👍
Carl Lauter MD
2488912451 mobile
2485517330 office
2585517268 fax
Carl.lauter@beaumont.edu
Sent from my iPhone
LikeLike