Today the FDA published its review of the phase three clinical trial for Moderna’s COVID-19 vaccine. The results were almost identical in both efficacy and safety to what Pfizer presented a week ago. There were, however, some interesting points that came from the review.
Overall efficacy was very similar to Pfizer’s data. As compared to the AstraZeneca vaccine, where infections continued to occur in the vaccine group, the Moderna vaccine’s efficacy resembled Pfizer’s vaccine, where COVID-19 infections in the vaccine group essentially stopped early on after the vaccine was received.

The trials were somewhat different, however, because this was a one-to-one comparison with about 15,000 people in both the active and control arms. In addition, the Pfizer vaccine’s second dose is given three weeks after the first dose while with the Moderna vaccine’s two doses are four weeks apart. While this timing difference likely has little effect in the long-term immune response, it does allow a closer examination of the first few weeks after the initial shot.
The analysis of these first weeks shows that the vaccine was highly effective beyond 14 days after the first dose. The vaccine was 50% effective for the first two weeks after the initial shot, but beyond that was almost as effective (92% reduction in infections) as the result after the second dose (95% reduction). Both these vaccines work fast and essentially stop all clinical infections.

Safety was also excellent and similar to Pfizer’s vaccine. Overall, the most frequently reported systemic adverse reaction was fatigue, reported by 68.5% of vaccine recipients and 36.1% of placebo recipients. After any dose, significant fatigue was reported by 9.6% of vaccine participants and 1.3% of placebo recipients. The median duration for fatigue in vaccine recipients was two days after any dose. The highest rates of fatigue were reported by participants 18 to 64 years of age after the 2nd dose, with 67.6% reporting any fatigue, 10.6% reporting significant fatigue.
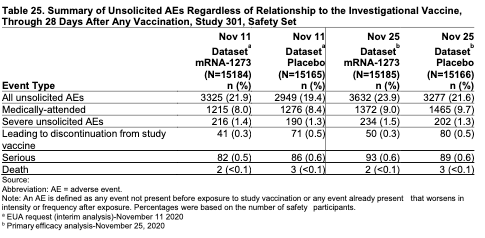
When examining issues that have popped up around the Pfizer vaccine, there was a small imbalance in the number of participants reporting Bell’s Palsy (facial paralysis), with three vaccine recipients and one placebo recipient. All the individuals had significant pre-existing health problems, including a stroke in one of the vaccine recipients that occurred about 40 days after the vaccine second dose. Therefore, an assessment of any relation to the vaccine isn’t clear, but the FDA will recommend surveillance for cases of Bell’s Palsy with deployment of the vaccine into larger populations.
There were no other notable patterns or numerical imbalances of adverse events between vaccine and placebo groups including other neurologic, neuro-inflammatory, and thrombotic events, that would suggest a causal relationship to the Moderna COVID-19 vaccine.

There were no systemic allergic reactions occurring within 30 minutes of vaccination, and a review of all adverse events that occurred within 30 minutes of vaccination demonstrated comparable rates across study groups (0.6% vaccine, 0.6% placebo), and none of the events reported in the vaccine group was considered serious.
The safety and efficacy of the Pfizer vaccine were recapitulated in the Moderna vaccine. It is likely this vaccine will be approved just like the Pfizer vaccine and provide many additional doses of vaccine. If asked to pick between the two, I would take either and opt for the one that was available first!
Why the temperature storage differential?
If both vaccines are so successful after one dose, and there is a shortage, why not split doses until adequate supplies are available?
LikeLiked by 1 person