Johnson and Johnson’s coronavirus vaccine is submitted to the FDA.
The pharmaceutical company has applied to the FDA for emergency use authorization for its one dose coronavirus vaccine. Their application came after the publication of their successful clinical trial this week in the New England Journal of Medicine. It showed that the vaccine was 66% effective at reducing coronavirus infections after a single dose.

It is likely that the vaccine could be approved during the FDA advisory committee meeting on February 26th, which could make it possible that the vaccine would be available sometime in March.
Again, I feel it is fine to approve this as its single dose vaccine in the short term, but I believe it will likely be much more effective if used with a second dose.
The government had purposely overestimated the amount of vaccine to assure adequate quantities necessary to immunize the nursing home population. The numbers were based on an estimated nursing home population and the concept that there would be one care person for every resident in the nursing facilities.

Several things made this estimate a marked overestimate of how much vaccine was necessary. First, it turns out that the nursing home population has shrunk dramatically in the past year due both to losses from COVID-19 and the fact that many individuals have moved out of nursing homes to avoid potential infection with the virus.
Added to this, unfortunately, fewer nursing home caregivers agreed to take the vaccine. The excess vaccine will be redistributed to individuals in a number of states like Illinois which are far behind in their vaccination programs.
The FDA approves the first home test kit to diagnose COVID-19.
U.S. public health experts have called on the federal government to approve and fund cheap and fast at-home Covid-19 tests to help stop the spread of COVID-19. This week the FDA approved the first test from Ellume, an Australian company. In addition, the Biden administration this week announced a $231.8 million deal to ramp up production of the test.
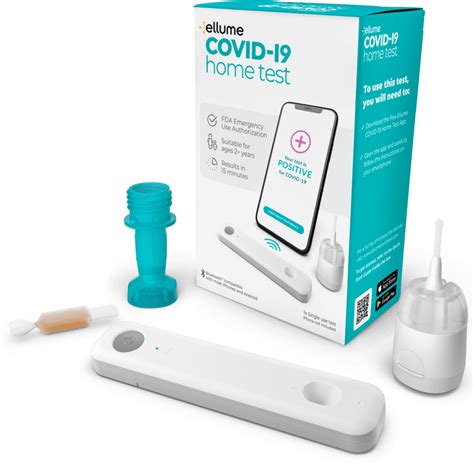
Unfortunately, the experts’ response was unenthusiastic. “It is priced at $30, far too expensive for most Americans to conduct frequent, regular screening at home. And the test is linked to a smartphone app that requires some tech savvy and steps” said Michael Mina, an assistant professor of epidemiology at the Harvard T.H. Chan School of Public Health.
Another expert dismissed it as, “a spit in the ocean. It’s not that home testing with a 15-minute turnaround time isn’t a good idea,” they said, “it’s just that the rollout of this initial kit is too little and too late, and the test too expensive and complicated, to help extinguish the raging pandemic.”
Hey Jim–Is J&J entry truly a preventative “vaccine”? If it’s 66% effective reducing Covid infections, does that mean: it helps two out of three Covid infected people w symptoms? Or: it helps reduce two-thirds of the symptoms? And if it’s only 2/3 effective ONCE someone has Covid, isn’t “vaccine” a misnomer? Thanks and take care!
LikeLiked by 1 person
Reduces all infections by 66% and serious infections by more than 90%. So protects against infection but effectively preventing disease.
LikeLike
Now that many of us are getting vaccinated, I would like science-based information as to what that means in getting back to some kind of normal life. Can I have an in-home dinner with friends that have also been vaccinated? Can I go into stores more safely with a mask and social distancing? Can I be with my grandchildren inside, if we all wear masks?
Could you provide updated information regarding this issue on a regular basis, as I know it will evolve with time based studies?
LikeLiked by 1 person
I think you should be safe and able to be inside with ANYONE who is vaccinated with out risk to either party.
without a mask, there is a very, very minimal (!) risk to unvaccinated children from you, if you happen to be in the 5% who get infected after being vaccinated. But since COVID remains a minimal illness in children, you and their parents can decide on that concern. You personally should be good to go though, and your mask is basically to prevent others from the remote risk you are infected!
LikeLike
Dr. Baker, Can you explain why the FDA is waiting until Feb. 26th to look at the data? What could be more important than getting this vaccine launched? What could the FDA be doing that is more important than reviewing this data?
Thanks, M.K. Toumajian
>
LikeLiked by 1 person
There are about 10,000 pages of data in these applications. They review all the adverse events, the statistical inference and the manufacturing to make sure what the company is saying is accurate. While I do not expect problems, there have been instances where the data was “fudged” or toxicity issues were not documented or addressed. So, as painfully long as it seems this is the last chance to catch any problems before the vaccine is unleashed on the public.
LikeLike