Attempts to develop vaccines for coronavirus variants highlight the differences in vaccine technology for COVID-19 and the implications for manufacturers.
An article in today’s Wall Street Journal highlights efforts by Moderna and Novavax to create new versions of their SARS-CoV-2 vaccines to specifically address Brazilian and South African virus variants. I and other scientists believe that the current RNA vaccines will be sufficiently effective against these variants, but this is not clear for Novavax’s vaccine. In any case, it is a useful endeavor to look at the effort and timing required to make a new or combination vaccine if a new form of coronavirus emerges.
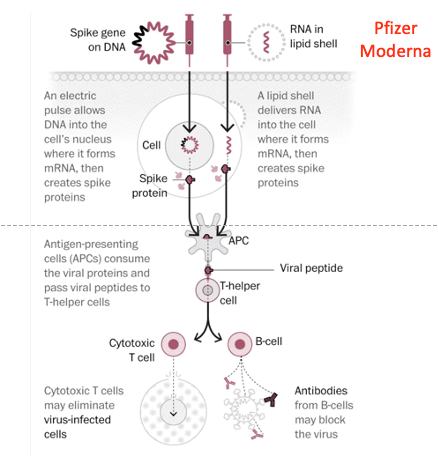
While the Moderna vaccine is already in wide use and highly effective against all currently circulating SARS-CoV-2 strains, these strains present a much bigger concern for Novavax’s vaccine. It appeared to be much less effective in their clinical trial in protecting against the South African variant. While some have dismissed this as simply the later timing of their vaccine trials, at a point when this variant was more prominent, the difference in the vaccines may also be important in this potential deficiency.
The Moderna vaccine uses the RNA for the specific regions of the SARS-CoV-2 spike protein involved in binding to lung cells to infect people. Changing the vaccine simply involves changing the RNA sequence to correspond to the new variant. This can be done in a few weeks, with manufacturing ready to start immediately. Moderna started this process after the first of the year and already has vaccine prepared.
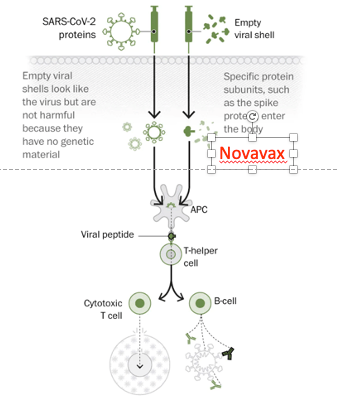
Novavax uses more traditional technology. They take the gene for the spike protein and place it into an insect virus. They then have to produce the virus and use it to infect cells (since it is an insect virus they infect moth cells!). The moth cells make the spike protein, which aggregates in the cells. The protein then needs to be isolated from moth cells and purified to be put into the vaccine. This is a more complex process and can take months rather than weeks to accomplish. They hope to begin trials in the middle or later months of this year.
Given the speed at which SARS-CoV-2 vaccine research has progressed, one has to wonder where Novavax fits in. There were delays in their clinical trials in the U.S. so the initial application to the FDA for approval of their standard COVID-19 vaccine in the U.S. is not expected until late March or April. This means approval is not likely until May, a time-point when the U.S. will likely have enough supply of the currently approved vaccines to treat the adult population.
If it takes six months just to develop an alternative vaccine before testing can start, Novavax may be even further behind in introducing their variant formulation. The speed and ease of production and modification inherent in the RNA Vaccines have transformed the COVID landscape, and it may just be that Novavax and its traditional technology cannot keep up.