
I have been hoping for a rational drug treatment for COVID based on science, not wishful thinking. One may be at hand!
Remdesivir is one of the first drugs that appears to be useful to treat Coronavirus infection. It is an antiviral drug, which means it specifically inhibits an activity that allows the virus to survive and replicate (make copies of itself). What is most impressive about Remdesivir is that it has a well-defined, specific mechanism of action. We know exactly what it does to the virus! This is a huge advantage when compared to several other drugs, like chloroquine, where the effect on the virus is ill defined.
How does Remdesivir work? Antiviral drugs in general can be considered poisons that are specific for virus biological activities, but don’t affect normal biology in humans. To develop an antiviral drug, one must first understand how the virus works. From this understanding, unique activities of the virus can be identified and targeted.
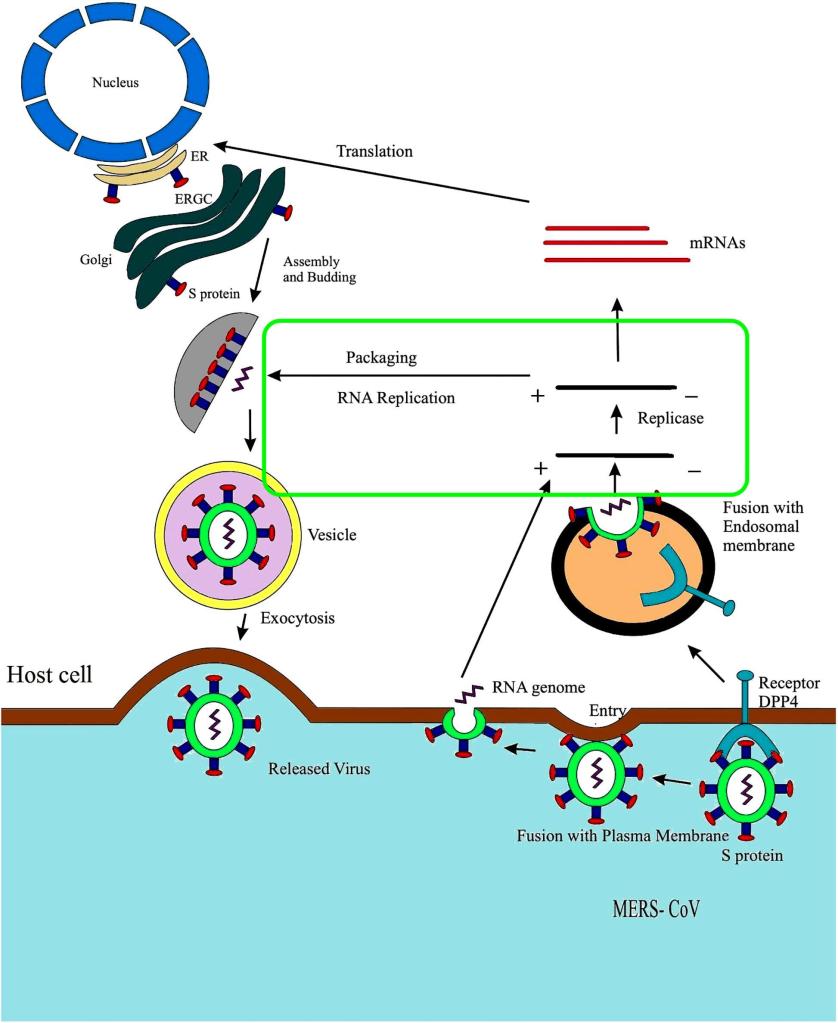
Remdesivir targets a unique activity of the virus. Because coronavirus is a RNA virus, it must make RNA copies of its own RNA to reproduce (make additional copies of the virus). In humans our genetic material is based on DNA. We only make RNA from DNA and only when we make protein. Humans never make a RNA copy of RNA, so the enzymes and other molecules that do this are unique to the virus. Thus, inhibiting the ability of the virus to make RNA copies of itself inhibits the virus reproduction but does not harm humans.
The way that remdesivir blocks RNA copies is by substituting itself in place of a normal nucleotide in the new RNA chain. Remdesivir then causes the polymerase enzyme, that makes the RNA copy of RNA, to stop working. This prevents the production of an RNA copy so that no new virus can be produced.

Remdesivir is not perfect. It can be overwhelmed by an excess of normal nucleotides, and when this occurs, some virus is produced. The key is that overall viral production decreases significantly, and this gives the patient’s immune system a chance to destroy the virus and the viral producing cells.
Remdesivir was originally developed for Ebola, another RNA virus where it worked by the same mechanism. It has undergone human safety studies, and an unblinded study published in the New England Journal of Medicine showed promise, albeit it was not fully convincing. Controlled, blinded studies are currently being conducted and if the drug works it could be approved by mid-May.
Gilead sciences, the company that makes the drug, believes they could treat 100,000 patients by the end of May and several million by the fall. If the drug truly prevents death in infected individuals, it could be a crucial turning point in the COVID-19 fight and facilitate our efforts to normalize activities.
One thought on “Remdesivir to the Rescue?”