
- Most COVID-19 antibody tests available to use outside a laboratory involve “lateral flow assays” named because they use diffusion of serum antibodies laterally in a paper strip.
- Blood or serum is placed at one end of the strip, and if it binds to a gold nanoparticle coupled to coronavirus proteins the nanoparticle is captured at a specific point in the paper.
- The nanoparticle concentrated in the strip reflect red light causing a bright red band. Negative sera do not make a band.

Assays that measure antibodies to specific proteins, like virus proteins, used to be very cumbersome procedures. Thirty years ago, they involved machines that took up an entire room, and generated radioactive waste with glass vials that created disposal issues. These tests were run in large laboratories and could possibly do one hundred samples a week. The Nobel prize for developing blood tests using antibodies was awarded to Dr. Rosalyn Yalow in 1977 (the year before I graduated from medical school!). This is still a young field.
Remarkably, in the past 20 years, immunoassays (assays tests using antibodies) have evolved to the point that they fit in your hand, don’t require radioactivity or other complex technology, and can give you an answer in minutes instead of weeks. In fact, many of us know about disposable immunoassays in our own home from pregnancy tests, one of the first technologies developed (and an acute need to know).
My discussion will focus on assays that are available to test for COVID antibodies that, like the pregnancy test, are likely used by individuals outside of complex laboratories, without a knowledgeable physician (pathologist) to oversee their use.

Most assays for COVID-19 antibodies use what’s called a lateral flow device. This is a remarkably simple technology, where either blood or serum is placed in a well on a piece of paper. A fluid called buffer is added that contains gold nanoparticles coupled to COVID-19 proteins. If there are antibodies to COVID-19 proteins in the blood, they bind to the proteins on the nanoparticles. This mixture then diffuses through the paper, until it meets another antibody that is coupled to the paper. It recognizes either human IgM or IgG antibodies and binds them and the COVID gold nanoparticles to the paper.
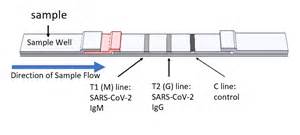


Reading the assay simply involves looking for lines. A line at the M position means there is IgM against COVID. A line at the G position means there is IgG antibodies against COVID. A line at the C or control position has to be present to show the assay worked.
Actual results

Results: 13;IgG and IgM are positive. 14 and 15 are negative. 16; borderline positive for IgG. 17 Positive for only IgG. 18; positive for only IgM. All the tests are valid since a control like is present in all lanes. A representation of all possible results is presented below.
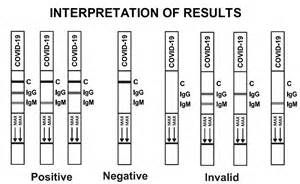
There are variations of this assay; some look only for IgG or IgM, other use two lanes, one for IgG and one for IgM. But the concept is the some for most of these assays.
It all seems very easy and it pretty much is. However, many factors can make the result unreliable. Some reliability issues involve the assay itself, some involve the operator and some involve the blood sample. Tomorrow I will discuss these problems and how they can be avoided.
An additional note; The physics behind this are very interesting and deserved your attention. It involves a phenomenon called surface plasmon resonance, that relates to the nanoparticle size of the gold. This is why the material it appears red and not gold and it is in itself an interesting story. Nanoparticle gold “stains” were used on stained-glass windows in the Middle Ages to give vibrant red colors that don’t fade over time. It is worth a review at this website!
One thought on “How COVID-19 Antibody Tests Work”