
As discussed in the blog post yesterday, the COVID-19, lateral flow antibody test is a remarkably simple, very stable and consistent platform. It requires no machine to run, it can be stored at up to 86°F for up to a year, and as long as the reagents are unused it gives very consistent results. Despite this, these assays have several issues that can cause them to fail.
The test failures falling to three general areas the first is sample preparation and the second operator error in the third problems with the test itself. I will go through and give specific examples of each type of failure and then have some general comments about the meaning of the essay at the end.

Sample issues
Timing. Antibody is part of an immune response to viral infection; therefore, it is not present immediately when the infection occurs. In fact, as is shown in this figure, antibodies only begin to appear between five and 14 days after the infection starts. You can see that IgM antibodies show up as a first defense, however these are not as strong as the IgG antibodies. The obvious problem here is that if you don’t wait long enough after the infection starts the antibody test will be negative. This is particularly important when looking at time frames where infection numbers are increasing rapidly; you need to remember is the antibodies tend to trail two weeks behind the actual infections.

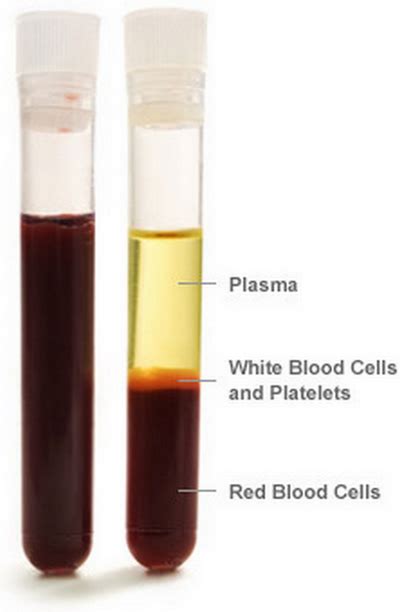
The type of sample. The antibodies are in the fluid component of the blood (called plasma or serum). If using the plasma or serum, where the fluid has been separated from the blood cells, it is easy for the antibodies to diffuse into the paper assay. May people prefer to use drops of blood obtained from a finger stick, because it is easier to do and there is little residual blood waste, and you do not need a phlebotomist (person to draw your blood through a needle). However, with whole blood the fluid has to move beyond the blood cells into the device. If the blood clots, it can prevent the fluid from entering the device giving a negative result. Thus, the blood cells themselves may alter the reliability of the sample.
Handling issues. How samples are obtained may also be an issue. If the blood is allowed to stand and or gets heated it will destroy the antibody. For example, if blood samples are left in the back of a hot automobile they can heat to a point where the antibody will be broken down. The red cells can also break down (hemolyze) and their contents may make the assay unreliable. Thus, care must be taken that samples are used rapidly after their obtained or that the fluid component is isolated and frozen to make sure that the antibodies do not degrade.
Operator problems
Not using or mixing the materials properly. Too much or too little blood sample can give false negative results either because the gold particle reagents are diluted or there’s not enough antibody to assay. Adding too little buffer will provide too few nanoparticles and again will give you a negative result. Operators can add the reagents to the wrong well or not add them in the proper sequence. Some people have a problem reading the test, particularly with red-green color blindness (cannot see the red line). In addition, even if an operator reads the test correctly, they may misinterpret what the lines mean. These many issues argue it is best to have an experienced individual perform the test.
Test design and manufacturing concerns
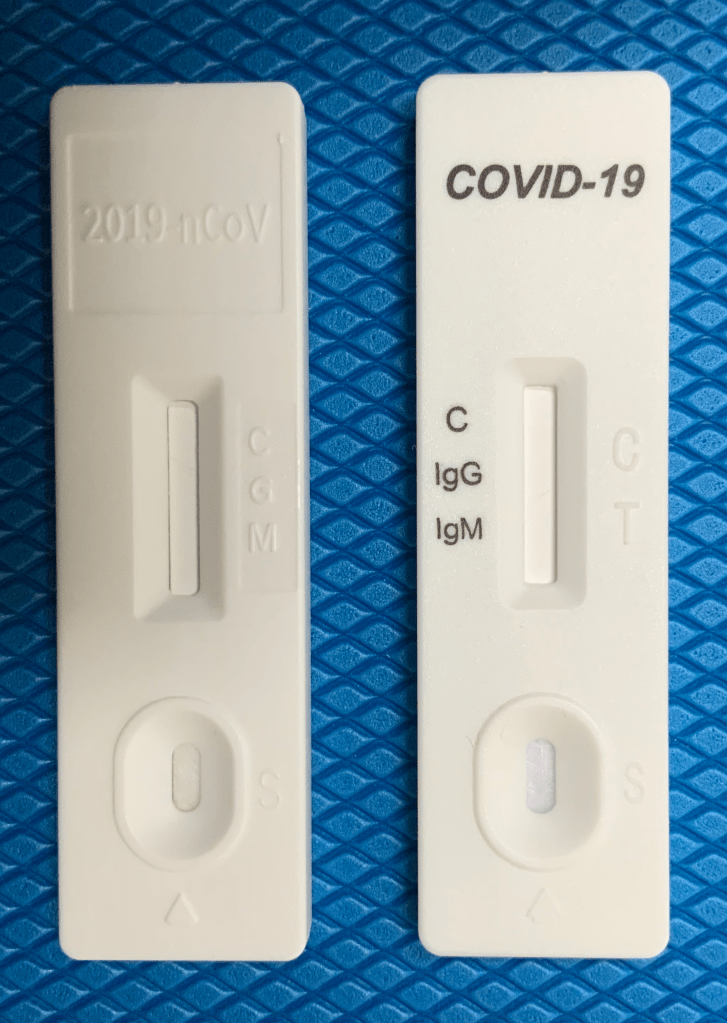
Finally, the test itself may be problematic. It may have been poorly manufactured, so the markings do not line up with the red (reagent) lines. Another issue can be seen in this picture were the markings on one device are simply embossed on the white plastic shell. This particular test would be difficult for an operator to read the bands correspond to IgG and IgM. On this particular essay, people also confuse the C (control) line with the G (IgG) line. Some tests have faulty reagents including the gold nanoparticles the COVID-19 proteins coupled to them. This again will give false negative results. The test can also have been stored improperly and not function.
The largest issue with all of these assays, even if they are done correctly, is what do the results mean!
Finding antibody does tell you that you have been exposed to and infected with the COVID-19 virus. This is of value; however, unlike a pregnancy test, where a yes/no answer is adequate, immunity is a not an all or none event. We do not definitively know if just the presence of antibody tells you that your protected from future or recurrent infection. It also gives no idea to the degree or amount of antibody or immunity you have, or whether it functions to neutralize virus. Those require much more complex test.
We need to do prospective, controlled studies to determine if the presence of antibody shows protection from infection. We are planning such a study in healthcare workers with occupational exposure to COVID-19. Only after studies like this are completed will be we be able to truly say that the presence of antibody documents protection against COVID-19.