The FDA today took action to clear up the mess in COVID-19 blood testing. You might remember that blood tests for COVID-19 were allowed to enter the US under a special emergency use approval (EUA) called Part D.

Under this special exemption, a test could be imported and sold on a “promise” that the manufacturer would file data showing efficacy at a later time. There was no time limit placed on this requirement, however, meaning a test could be imported and sold for an indefinite period of time without ever providing data it actually worked.
The agency also provided performance recommendations regarding specificity and sensitivity for test developers. Specificity measures how often the tests produce an incorrect result, while sensitivity measures how often the test shows a correct positive result
This attempts to close the loophole that the FDA created when it allowed importation and sales of devices under part the of the emergency use authorization. While there had to be a disclaimer on the device suggesting it was not used for human diagnostic applications, many people ignored that label. Large sales of untested devices, mainly produced by Chinese companies, flooded the US market.
At one point it was estimated that around 150 different companies were attempting to sell tests in the United States with prices that ranged up to $300. This created such a blizzard of bad press the Chinese government (to their credit) stepped in and prevented export of blood tests that were not approved by China’s FDA. Despite this, sales of unapproved devices with no efficacy validation have continued.

In my opinion the FDA should never have allowed sales to occur under Part D without full efficacy and use testing. The new rules are better as they require companies to file data packages with the agency within 10 days. However, it’s not clear to me that this will prevent people from simply supplying an inadequate data package to the FDA then continuing to sell under the Part D authorization.
The best thing for the FDA to do would be to require any COVID blood test to get full EUA approval before it can be sold in the United States. While this will restrict testing options initially, in the long run it will prevent unscrupulous and unauthorized devices from flooding the US market.
Since many of these devices don’t appear to work, I believe that limiting their import and use is no great loss. There also are now multiple devices that have achieved full EUA so there are options for individuals to use to test for COVID antibodies.
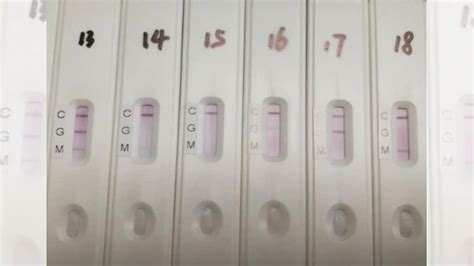
As we allow people to return to normal lives, if we use blood testing in this process, we first need to make sure that the tests are accurate. After that we can determine whether the presence of antibody indicates protection from COVID.
I would love to hear your thoughts on Mikovits!
LikeLike
They keep pulling the video off YouTube, but when I saw it I wasn’t surprised. She is an anti-vaxxer who is now taking advantage of this difficult situation to spread misinformation. As polite as I can be on this.
Jim
LikeLike