Today the journal Lancet published two studies of new vaccines for COVID-19. The studies were interesting in that both involved adenovirus virus systems to induce immunity. One study provided the data on a chimpanzee adenovirus COVID-19 vaccine (ChAdOx1) from Oxford University and the AstraZeneca Corporation. The other involved a human adenovirus 5 system (Ad5) from CanSino Corporation in China.


Both of these systems use an adenovirus (a common cold virus) that has been genetically engineered to produce SARS-Cov-2 proteins. The viruses infect the vaccine recipient and induce an immune response that includes the SARS-CoV-2 proteins. It is hoped that the immune response will be able to prevent COVID-19.
These are two of the 250 vaccines that are in development for COVID-19. These vaccines are also two of the first to reach clinical trials. Both trials are large and well controlled with placebo arms. These early trials are done mainly for safety, but also include measurements of the immune response to the COVID-19 viral proteins.
There are several similarities between the results from these two vaccine trials. Both show evidence of significant antibody responses against the ARS-CoV-2 virus proteins. Both vaccines also induced antibodies that could neutralize the virus in about half of the recipients of the vaccines. In addition, the vaccines showed some evidence of cellular immunity; producing T lymphocytes (T Cells) made cytokines (interferon) that have antiviral activity against COVID-19. The ability to induce an immune response with both of these vaccines appeared stronger than the recent results from the Moderna vaccine.
Another major similarity is that there were significant adverse events associated with both vaccines. Approximately 70% of people getting the higher dose of the CanSino vaccine and 70% of the people after the Oxford vaccine had significant systemic events side effects compatible with a severe flu like illness. These included fever, sore muscles, nausea, headache and fatigue (see figures below). In the Oxford trial the symptoms were much worse than those with a meningitis vaccine given as a comparator. However, in neither trial was there an adverse event so horrible that it caused the trial to be stopped. It is likely that a lower dose of Ad5 will be used in future trials, and two doses of the chimp adeno will be needed.

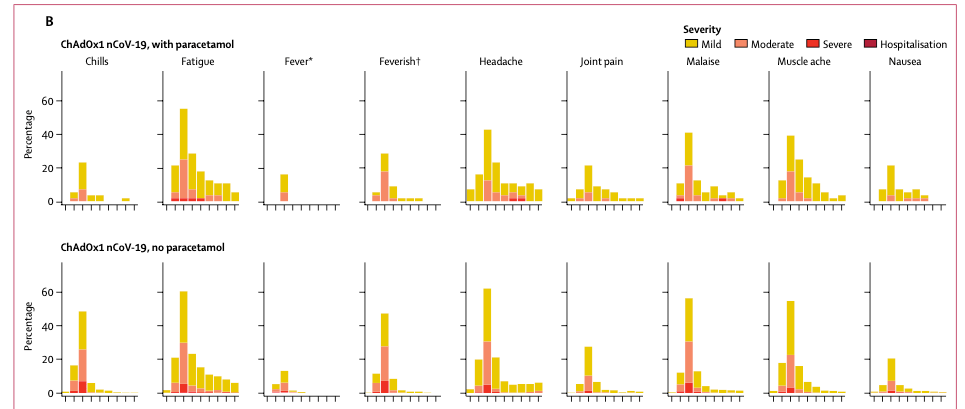
There were several significant differences to these vaccines. Ad5 is a human virus and many people already have immunity to this virus which could reduce the effectiveness of that vaccine. In their trials older subjects had less side effects from the vaccine, and that correlated with prior infection with Ad5. This suggests immunity to Ad5 might be an issue.

Of interest, the chimp adenovirus was used to avoid using the human adenovirus. The side effect profile was much the same, however. It also appeared to require two “shots” to achieve optimal immunity as compared to one administration with the Ad5 vaccine.
Both vaccine companies had previously announced study results in press releases. The results in both studies largely confirm what was already previewed. In a comment on the chimp vaccine Adrian Hill, head of Oxford’s Jenner Institute, told Bloomberg, “We are seeing very good immune responses, not just on neutralizing antibodies but of T-cells as well. We’re stimulating both arms of the immune system.” He also noted the increased immune response vs. Moderna’s rival vaccine.
In summary, the two vaccines induce immunity but cause significant side effects. Again, there is no guarantee that either will work, but based on these results both are safe enough to move forward to larger trials. Whether they will be safe enough to give to large, diverse populations remains to be determined.
One thought on “Two new COVID-19 vaccine trials in the Lancet”