On Friday, the medical Journal Lancet published the results of the first clinical trials involving the Russian COVID-19 vaccine.
As background, the Russian vaccine (nicknamed Sputnik V after the famous satellite) is made from a recombinant virus that causes human colds upper respiratory infections (adenovirus) that has been genetically modified to produce the spiked protein from the human COVID-19 virus SARS-CoV-2. Two different human types of adenovirus (recombinant types 26 and 5) were produced with the spike protein, and preliminary testing in humans examined whether these viruses could produce immunity to the COVID spike protein and whether a frozen versus a freeze-dried formulation was better.
Specifically, the study was conducted between June 18 and Aug 3, 2020 and enrolled 76 participants (38 for each virus). In each study, nine volunteers received rAd26-S in phase 1, nine received rAd5-S in phase 1, and 20 received rAd26-S and rAd5-S in phase 2. Therefore, the number of participants was limited, and all were adults between the ages of 18 and 60 years of age. The vaccine was injected into the arm of each recipient, and in the second study a booster immunization was administered 21 days after the first.
Both vaccine formulations were safe and well tolerated. The most common adverse events were pain at injection site [58%], fever [50%], headache [42%], lethargy [28%], and muscle and joint pain [24%]. Most adverse events were mild, and no serious adverse events were noted.
All participants produced antibodies to SARS-CoV-2 glycoprotein, as well as evidence of cellular immunity. The antibody titers were as strong as those from patients who had recovered from COVID-19. The study ended at 42 days, so no long-term results could be identified.
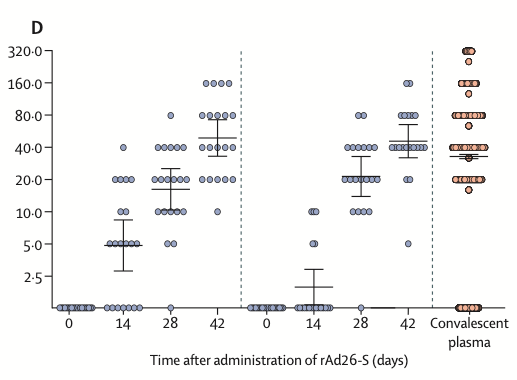
The most remarkable result from these trials is how normal they appear. The results appear identical to that seen with COVID-19 vaccines based on recombinant adenovirus vaccines developed in the Western world. These included an adenovirus type 5 (Ad5) vector-based vaccine (CanSino Biological/Beijing Institute of Biotechnology, China), an Ad26 vector-based vaccine (Johnson & Johnson, USA), and a vaccine containing a chimpanzee adenoviral vector (AstraZeneca/University of Oxford, UK). The Russian trials are even registered with the US FDA clinical trial site, ClinicalTrials.gov, and identified as NCT04436471 and NCT04437875.
Why did the Russians produce two vaccines based on different adenoviruses? The main issue that can limit use of vaccines based on recombinant adenoviruses is pre-existing adenovirus immunity in the human population. After vaccination with an adenoviral vaccine, immunity develops to the COVID spike antigen but also to the adenovirus itself.
While immunity to the recombinant adenoviruses did occur after vaccination with rAd26 and rAd5, development of antibodies to the target antigen (SARS-CoV-2 glycoprotein S) was not affected. Moreover, neutralizing antibodies to rAd26 did not neutralize rAd5 when serum samples from vaccinated volunteers were obtained and analyzed 28 days after immunization (and vice versa). Despite this, use of rAd26-S as a first shot followed by rAd5-S as the second “booster” shot might be a more effective way to elicit protective immunity.
The results suggest that the Russian vaccine is legitimate and very similar to several similar vaccines based on similar technology in the Western world. The biggest difference is that the Russian vaccine is being given out without having completed clinical trials.
Time will tell whether it works as well as as the Western counterparts.