The FDA today released its internal review of the Pfizer/BioNTech request for emergency use approval of their COVID-19 vaccine. The document, which stretches 53 pages, is available for download and remarkably complete. It reveals that the vaccine met all efficacy and safety milestones set by the agency and strongly suggests the vaccine will be approved by the review committee.
Remarkably, no major surprises or concerns were identified. This is unusual for such a large and comprehensive (44,000 people) data set. However, there were a few insights that had not previously been made known. Most make the data, if anything, more compelling.
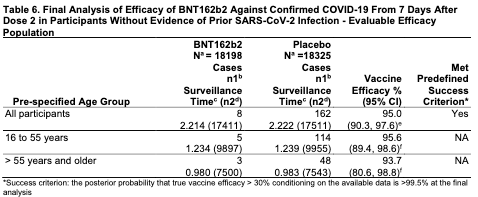
The top line again is that the vaccine is over 95% effective regardless of age or ethnic group.
Also, there were very few adverse events that were considered significant, however many of the subjects who actually got the vaccine felt like they had the flu for a day or so after the 2nd injection. These symptoms were significant enough that many are now suggesting that vaccine recipients be clearly cautioned about what to expect so they aren’t concerned they are having an actual COVID-19 infection or that they are having an unusual response to the vaccine.
Here is a list of other new findings from the FDA Review.
1) There is an indication that the vaccine appeared to provide some protection 10 days after the first dose. While the vaccine is designed for two doses, 21 days apart, infections in the vaccine group started dropping 10 days after the first dose of vaccine. Fourteen days after the first dose, infections in the vaccine group essentially stopped. This was remarkable.
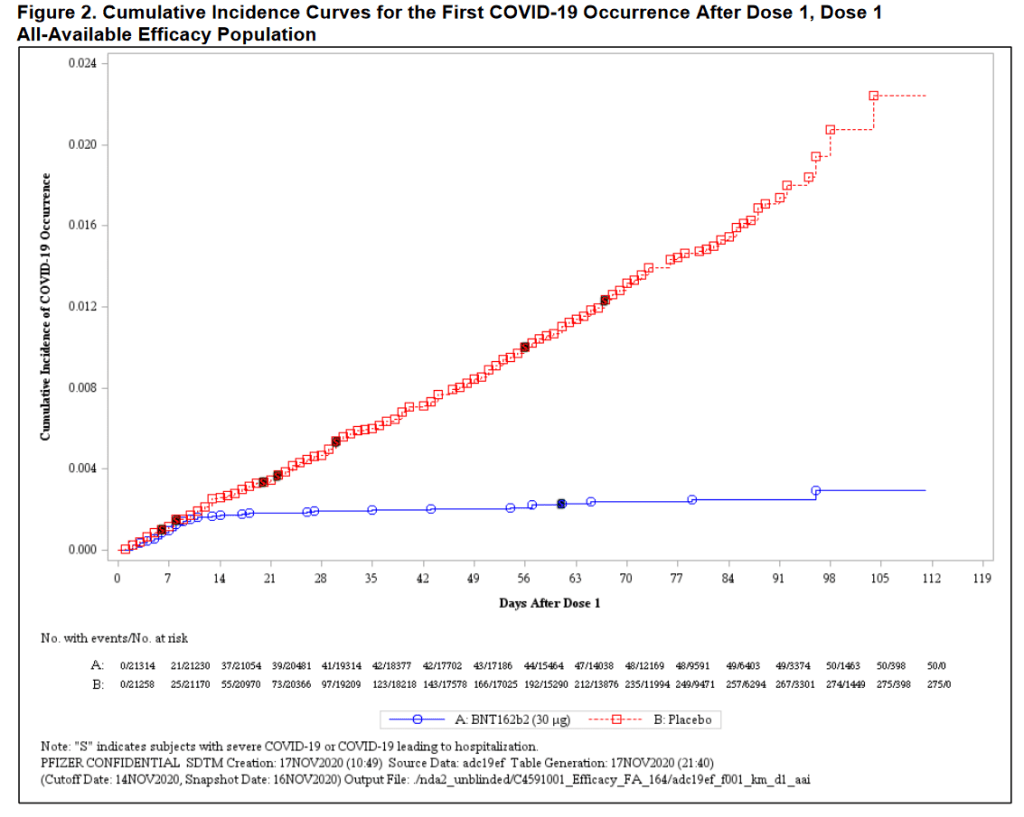
2) There were 9 severe infections in the control group, but only one in the vaccine group, providing evidence of prevention of severe disease. The one person with “severe disease” in the vaccine group did not require hospitalization or breathing support; even more reassuring.
3) In contrast, there was no evaluation of asymptomatic infection in this study. Therefore, it is possible that asymptomatic infections were not controlled to the same degree as those that produce symptoms.
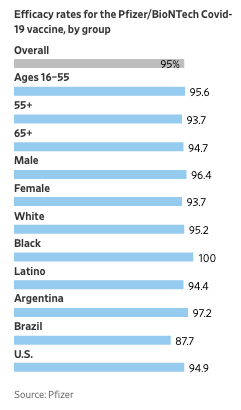
4) Efficacy was remarkably consistent regardless of any demographic factor or co-morbidity. If you are an obese person over 65 years old, this is your vaccine!
5) The data only support protection up to two months after vaccine therefore ongoing studies will have to be performed. Effectiveness in individuals younger than 15 was also not assessed, but given the broad effectiveness it might be presumed. Studies in younger children are beginning.
6) Data are limited to assess the effect of the vaccine against transmission of SARS-CoV-2 from individuals who are infected despite vaccination. Demonstrated high efficacy against symptomatic COVID-19 may translate to overall prevention of transmission in populations with high enough vaccine uptake.
7) The most common adverse reactions were injection site reactions (84.1%), fatigue (62.9%), headache (55.1%), muscle pain (38.3%), chills (31.9%), joint pain (23.6%), and fever (14.2%). Adverse reactions characterized as reactions to the vaccine were generally mild to moderate. Of interest, these reactions appeared to be less significant if the recipient was >55 years old.
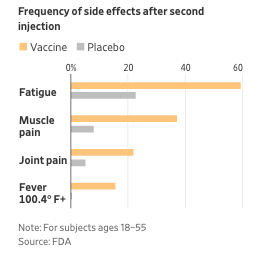
As stated above, this left many individuals with flu like symptoms especially after the second dose. It is appropriate that anyone getting the vaccine be cautioned and expect these side problems. A nurse who received the vaccine wrote that description of her symptoms in the Journal JAMA to help counsel people about what to expect from the vaccine.
Finally, there are the caveats of what the vaccine data can’t show at this point. Efficacy in pregnant or lactating women and immunocompromised individuals was not tested. There also is the possibility of some uncommon, late onset adverse reactions that hasn’t yet been seen. The only inflammatory problems in the study were four cases of Bell’s Palsy that were noted in the vaccine group alone, but this was not statistically significant versus controls.
There is also no evidence of the vaccine making the illness worse (in part because there were so few infections in the vaccine group!), and again there was very little data associated with individuals who had prior infection. This will require further study.
Regardless, from my review, I do not see anything that would prevent me from taking this vaccine or encouraging anyone else to take it. In fact, the risk benefit ratio of this vaccine compared to COVID-19 infection is so great that there is overwhelming reason too sign on!
Hoping you will comment on the allergic reactions in the UK NHS workers and how people with history of any serious allergies should be excluded from getting the Pfizer vaccine. https://www.reuters.com/article/health-coronavirus-britain-vaccine/uk-warns-people-with-serious-allergies-to-avoid-pfizer-vaccine-idUSKBN28J1DX
LikeLiked by 1 person
see todays post, just up.
LikeLiked by 1 person