This week in the Lancet, the data from the phase three trial of the Astra Zeneca/Oxford vaccine were published. The results had been previously announced in a press release and suggested that the efficacy was anywhere from 60 to 90%, dependent on the first dose of the vaccine. The data in Lancet finally clarify the efficacy and show the specific results.
The trial used a menigococcal vaccine and saline as controls and employed a number of unique analyses. Their primary endpoint was prevention of COVID-19 with “classic” symptoms, but they also analyzed “atypical cases.” Why this was split out was not clear. There was also an analysis of reduction in COVID-19 positive PCRs.
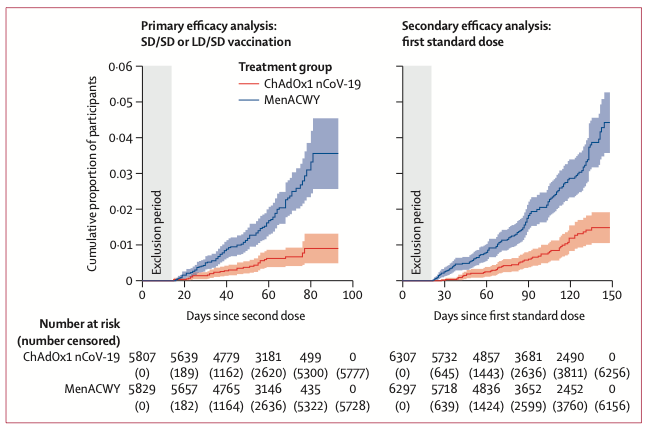
Trials were conducted simultaneously in Britain, South Africa, and Brazil (Table 4). Incidence of COVID-19 infection was twice as high in Brazil as in England (35 vs. 78 infections per 1,000 person years). Not surprisingly, efficacy appeared better in Brazil — 71% vs. 55%. When averaged, the overall efficacy of the two trials was 64%. However, when COVID-19 cases with atypical symptoms were added to the analysis, the overall efficacy fell to 58%. Reduction of positive COVID-19 PCR showed 46% efficacy.
Given these results, the overall efficacy of this vaccine is about 60%, which corresponds to the lowest figure given in the press releases.


It also appeared that Covid hospitalizations and severe disease were reduced. Unfortunately, there were very few cases, so the reduction was minimal and not statistically significant.
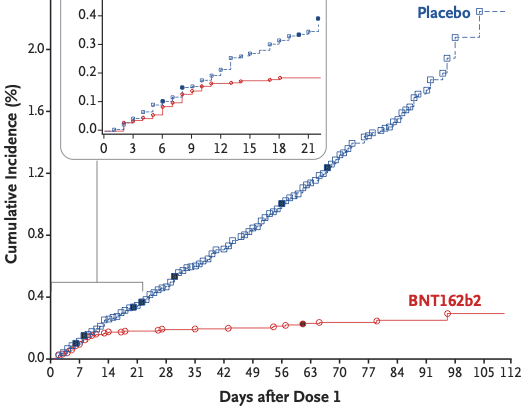
Probably the most interesting comparison is to the Pfizer vaccine, which is 95% effective. When comparing the first standard dose of both vaccines, the Pfizer vaccine essentially stops new infections 14 days after the first dose. The vaccine group in the AstraZeneca vaccine continues to accrue infections. This is a very significant difference.
AstraZeneca is pushing this vaccine hard. There are now proposals to mix dose this vaccine with the the Pfizer vaccine and even the Russian COVID-19 vaccine! This appears to be an effort to improve the marginal efficacy of this vaccine, especially when compared to the Pfizer and Moderna vaccines. If any of these efforts were to be done, completely new trials would have to be performed, and the results would not be available for many months.
Jim,
I am enjoying your FOBS (free-of-BS) briefs.
LikeLiked by 1 person
Thanks Lou. Hope you and your family are doing well. Jim
LikeLike
Please consider doing a post on the proper way to give IM injections. On TV all day today I am witnessing Covid vaccine injections during which the person performing the injection does not pull back on the syringe to insure that the needle has not entered a blood vessel before giving the injection. The news media and the locations promoted on TV should be alerted.
On Fri, Dec 11, 2020 at 8:15 PM Pandemic Pondering wrote:
> jbakerjrblog posted: ” This week in the Lancet, the data from the phase > three trial of the Astra Zeneca/Oxford vaccine were published. The results > had been previously announced in a press release and suggested that the > efficacy was anywhere from 60 to 90%, dependent on the fir” >
LikeLiked by 1 person