The FDA’s Vaccines and Related Biological Products Advisory Committee rejection of Pfizer’s proposed booster shots in all Pfizer vaccinated Americans was predicated entirely on the science. Despite the opportunity for Pfizer to make the case for vaccinating everyone 16 and older six months after they are fully vaccinated, and political support for this approach, the data were lacking.
Current studies have shown the Pfizer vaccine loses some protection from infection with SARS-CoV-2 over time, but the protection from serious infection and death remains impressive. There was a lively discussion by the committee, that also felt too little was known about the safety of a booster dose in younger people and pointed out that there was an absolute lack of data about the long-term efficacy of the booster shot. Some even commented that the process was rushed and wanted to see more data
All the data for the committee’s review came from Pfizer, and none was peer reviewed. In contrast, the independent data on decline of vaccine immunity over time was summarized in last week’s Lancet review co-authored by FDA vaccine scientists. The comments in that article indicated the FDA scientists believed that, “to date, no study has provided credible evidence of substantially declining protection from severe disease (with the Pfizer vaccine), even when there appear to be declines over time in vaccine efficacy against symptomatic disease.”
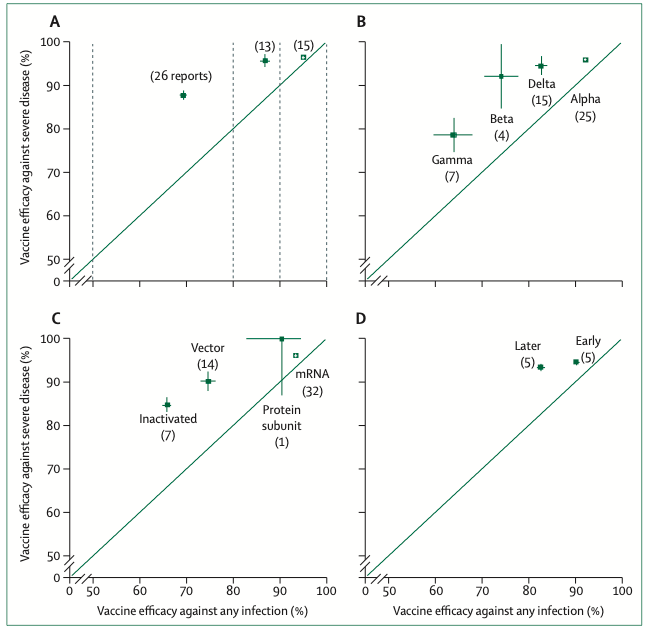
Review of published or informal reports of vaccine efficacy (with a 95% CI) in observational or in randomized studies (appendix pp 3–4) that gave results both for severe disease and for any infection. Plotted are inverse- variance-weighted means (and 95% CIs) of the reported vaccine efficacy (giving the number of studies contributing to that mean), subdivided by (A) Vaccine efficacy against any infection (50% to <80%, 80% to <90%, ≥90%). (B) Viral variant. (C) Type of vaccine (viral vector, inactivated SARS-CoV-2, adjuvanted protein subunit, or mRNA). (D) Studies reporting vaccine efficacy early (more recently relative to vaccination) or later (less recently relative to vaccination) during the follow-up of the same observational study.
Much of the data the administration cites in the need for booster shots is from Israel. The data is often not independently reviewed and is confusing. The Lancet review noted that “one study from Israel reported effectiveness against severe disease was lower among people vaccinated either in January or April than in those vaccinated in February or March, exemplifying the difficulty of interpreting such data.”
The most extensive, peer review report in the New England Journal of Medicine on the experience in Israel during the first three weeks of August 2021, just after booster doses were approved and began to be deployed widely, has suggested efficacy of a third dose (relative to two doses). However, the follow-up averaged only about 12 days! Obviously, a very short-term protective effect does not necessarily imply a worthwhile long-term benefit.
So, it appeared that the FDA and the review committee agreed on the limitations of the available data while the President and administration were moved by the anecdotal reports from Israel.
The committee did unanimously support authorization for booster shots limited to those over 65 and in higher-risk group, and they advised the FDA to include health care workers or others at high risk of Covid-19 exposure in the EUA. These groups appear to be most likely to have serious illness or death with the Pfizer vaccine.
The FDA will now decide on Pfizer’s booster requests, and then the CDC will weigh in. I doubt the CDC or the administration will go against the FDA, so if you aren’t over 65 or otherwise at risk, you won’t get a booster the week of September 20. In other good news, recent data from the CDC suggests that the Moderna vaccine does an even better job of protecting against hospitalizations [Among U.S. adults without immunocompromising conditions, vaccine effectiveness against COVID-19 hospitalization during March 11–August 15, 2021, was higher for the Moderna vaccine (93%) than for the Pfizer-BioNTech vaccine (88%) and the Janssen vaccine (71%)].
Therefore, most folks who got either MRNA based vaccine won’t need a COVID-19 booster for some time.